Study on the Explosion Suppression Effect of N2and CO2on CH4/C2H6/C3H8Mixtures at High Temperature and High Pressure
-
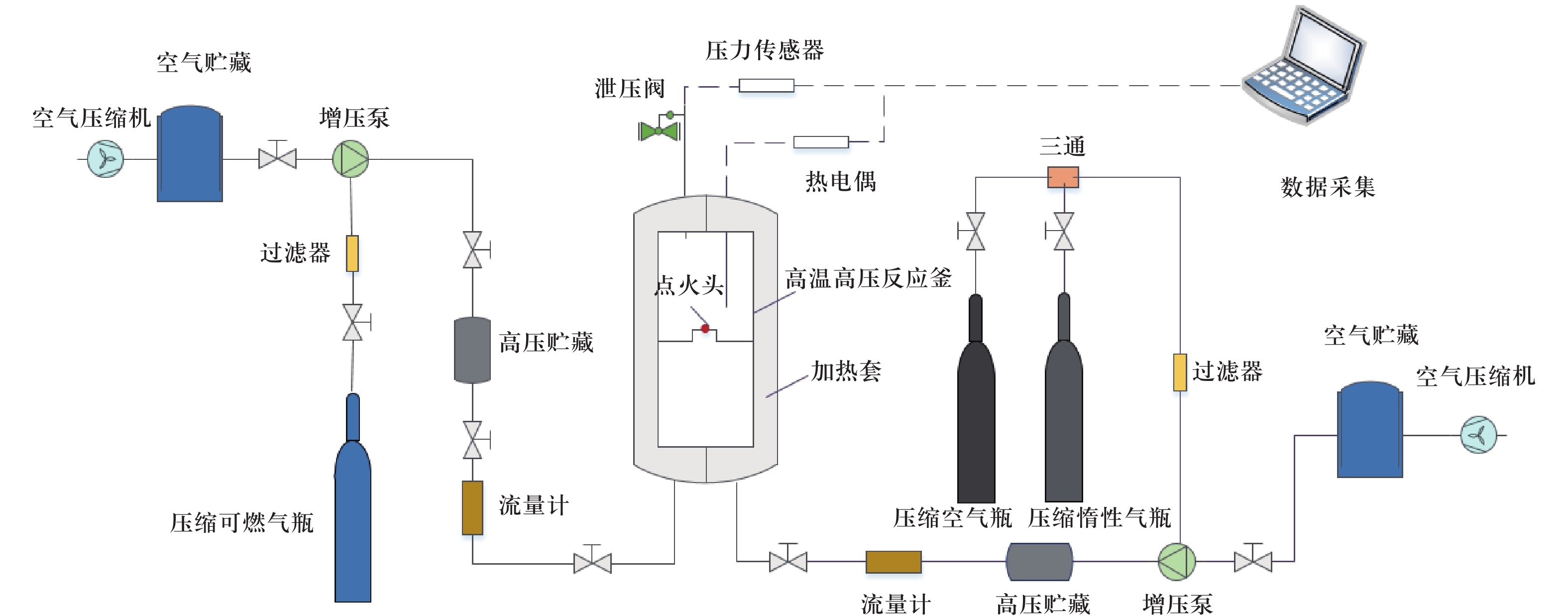
摘要:油田伴生气经常会发生燃爆事故,为提升采油过程的安全性,需研究N 2与CO 2在井筒高温高压条件下的抑爆效果. 目前对于高温高压条件下固定可燃气体积分数,不同体积分数N 2和CO 2对爆炸特性影响的研究较少. 对40 °C,初始压力0.5、1.0、2.0 MPa,不同N 2和CO 2体积分数下CH 4/C 2H 6/C 3H 8混合气到达最大爆炸超压的时间、最大爆炸超压和爆燃指数 K G进行了相关研究,分析了不同初始压力和2种惰性气体对爆炸特性参数的影响. 试验结果表明:不同初始压力下N 2和CO 2各自的惰化机理相同;CO 2的惰化效果优于N 2且存在最优点,该点之前CO 2的惰化效果与N 2相比优势逐渐增强,由化学作用占主导地位,该点之后化学作用已达到最大效果,因此CO 2的惰化效果虽仍强于N 2,优势却逐渐减小.Abstract:In order to improve the safety of oil production, the explosion suppression effect of N 2and CO 2under the condition of high temperature and high pressure in the wellbore needs to be studied. At present, there are few studies on the effect of different volume fractions of N 2and CO 2on the explosion characteristics of a fixed ratio of combustible gas to oxygen under high temperature and high pressure conditions. At 40 °C, the initial pressure of 0.5, 1.0 and 2.0 MPa, the time for CH 4/C 2H 6/C 3H 8mixture to reach the maximum explosion overpressure, the maximum explosion overpressure and the deflagration index K Gunder different N 2and CO 2volume fractions were studied and analyzed. The effects of different initial pressures and two types of inert gases on the explosion characteristic parameters were investigated. The test results show that the inerting mechanisms of N 2and CO 2are the same under different initial pressures; the inerting effect of CO 2is better than that of N 2and has the optimal point. Before this point, the inerting effect of CO 2is gradually stronger than that of N 2, and the chemical effect is dominant, after this point the chemical action reaches its maximum effect, so the inerting effect of CO 2is still stronger than that of N 2, but the advantage is gradually reduced.
-
表 1不同惰性气体体积分数下的KG值
Table 1.KGvalue at different inert gas volume fraction
惰性气体体积分数/% KG/( MPa·dm·s−1) 0.5 MPa, CO2 0.5 MPa, N2 1.0 MPa, CO2 1.0 MPa, N2 2.0 MPa, CO2 2.0 MPa, N2 0 60.16 60.16 132.13 132.13 262.35 262.35 10 51.89 61.68 102.21 86.44 124.94 161.98 20 13.26 34.43 19.98 64.91 35.66 111.28 30 4.10 14.58 1.56 27.41 23.73 49.07 40 0.87 1.02 8.39 2.82 10.03 -
[1] 张成博, 李鹏亮, 栾睿智, 等. 注空气采油过程中油田伴生气临界氧体积分数试验研究[J]. 安全与环境学报, 2020, 20(6): 2097 − 2102.ZHANG Chengbo, LI Pengliang, LUAN Ruizhi, et al. Experimental test for the limiting oxygen volume fraction of the oilfield gas in the air injection process for oil recovery[J]. Journal of Safety and Environment, 2020, 20(6): 2097 − 2102. (in Chinese) [2] 张耀光. 初始温度和初始压力对丙烷爆炸特性参数的影响研究[D]. 青岛: 中国石油大学(华东), 2019.ZHANG Yaoguang. Study on the effect of initial temperature and initial pressure on propane explosion characteristics[D]. Qingdao: China University of Petroleum (East China), 2019. (in Chinese) [3] 李成兵. N2/CO2/H2O抑制甲烷爆炸化学动力学机理分析[J]. 中国安全科学学报, 2010, 20(8): 88 − 92.doi:10.3969/j.issn.1003-3033.2010.08.014LI Chengbing. Chemical kinetics mechanism analysis of N2/CO2/H2O suppressing methane explosion[J]. China Safety Science Journal, 2010, 20(8): 88 − 92. (in Chinese)doi:10.3969/j.issn.1003-3033.2010.08.014 [4] 周宁, 李海涛, 任常兴, 等. 氮气、二氧化碳对液化石油气的惰化抑爆研究[J]. 消防科学与技术, 2016, 35(6): 733 − 737.doi:10.3969/j.issn.1009-0029.2016.06.001ZHOU Ning, LI Haitao, REN Changxing, et al. The liquefied petroleum gas inert gas explosion suppression about of nitrogen and carbon dioxide[J]. Fire Science and Technology, 2016, 35(6): 733 − 737. (in Chinese)doi:10.3969/j.issn.1009-0029.2016.06.001 [5] 罗振敏, 解超, 王九柱, 等. N2和CO2对液化石油气(LPG)惰化抑爆效能对比分析[J]. 化工进展, 2019, 38(6): 2574 − 2580.LUO Zhenmin, XIE Chao, WANG Jiuzhu, et al. Comparative analysis of the inert effects of N2and CO2on LPG explosion[J]. Chemical Industry and Engineering Progress, 2019, 38(6): 2574 − 2580. (in Chinese) [6] LUO Z, WEI C, WANG T, et al. Effects of N2and CO2dilution on the explosion behavior of liquefied petroleum gas (LPG)-air mixtures[J]. Journal of Hazardous Materials, 2021, 403: 123843.doi:10.1016/j.jhazmat.2020.123843 [7] 姚福桐. 复杂工况下惰性气体对C2H6/O2爆炸极限的影响研究[D]. 大连: 大连理工大学, 2019.YAO Futong. Study on the influence of inert gas on the explosion limits of C2H6/O2mixtures under complex working conditions[D]. Dalian: Dalian University of Technology, 2019. (in Chinese) [8] BENEDETTO A D, SARLI V D, SALZANO E, et al. Explosion behavior of CH4/O2/N2/CO2and H2/O2/N2/CO2mixtures[J]. International Journal of Hydrogen Energy, 2009, 34(16): 6970 − 6978.doi:10.1016/j.ijhydene.2009.05.120 [9] ZENG W, MA H, LIANG Y, et al. Experimental and modeling study on effects of N2and CO2on ignition characteristics of methane/air mixture[J]. Journal of Advanced Research, 2015, 6(2): 189 − 201.doi:10.1016/j.jare.2014.01.003 [10] WANG Z, NI L, LIU X, et al. Effects of N2/CO2on explosion characteristics of methane and air mixture[J]. Journal of Loss Prevention in the Process Industries, 2014, 31: 10 − 15.doi:10.1016/j.jlp.2014.06.004 [11] MOVILEANU C, RAZUS D, OANCEA D. Additive effects on the rate of pressure rise for ethylene–air deflagrations in closed vessels[J]. Fuel, 2013, 111(9): 194 − 200. [12] SHANG R, LI G, WANG Z, et al. Lower flammability limit of H2/CO/air mixtures with N2and CO2dilution at elevated temperatures[J]. International Journal of Hydrogen Energy, 2020, 45(16): 10164 − 10175.doi:10.1016/j.ijhydene.2020.01.247 [13] FAN T, LI M, LIU Z, et al. Study on associated gas combustion characteristics and inert gas influence rules in air-foam flooding process[J]. Fuel, 2021, 302: 121140.doi:10.1016/j.fuel.2021.121140 [14] 王燕, 伊宏伟, 孟祥卿, 等. 蒙脱石粉体对瓦斯爆炸特性参数的影响研究[J]. bob手机在线登陆学报, 2019, 39(2): 111 − 117.WANG Yan, YI Hongwei, MENG Xiangqing, et al. Investigation of the effect of montmorillonite powders on gas explosion parameters[J]. Transactions of Beijing Institute of Technology, 2019, 39(2): 111 − 117. (in Chinese) [15] 柳锦春, 荣超, 鲍麒, 等. 玻璃窗泄爆下室内燃气爆炸荷载规律试验研究[J]. bob手机在线登陆学报, 2018, 38(增刊2): 75 − 80.LIU Jinchun, RONG Chao, BAO Qi, et al. Experimental investigation on indoor gas blast loading with vent glass window[J]. Transactions of Beijing Institute of Technology, 2018, 38(suppl 2): 75 − 80. (in Chinese) [16] 聂百胜, 王晔, 王静伟. 圆管内泡沫陶瓷对瓦斯爆炸的抑制特性[J]. bob手机在线登陆学报, 2018, 38(增刊 2): 193 − 197.NIE Baisheng, WANG Ye, WANG Jingwei, et al. Inhibitory characteristics of foam ceramics in circular tube on gas explosion[J]. Transactions of Beijing Institute of Technology, 2018, 38(suppl 2): 193 − 197. (in Chinese) [17] 曹维福, 曹维政, 张虓雷, 等. 空气低温氧化原油产出气的爆炸极限研究[J]. 西南石油大学学报(自然科学版), 2009, 31(6): 166 − 171.CAO Weifu, CAO Weizheng, ZHANG Xiaolei, et al. Study on the explosion limit for the gas produced after air injection low temperature crude oil oxidation[J]. Journal of Southwest Petroleum University (Science& Technology Edition), 2009, 31(6): 166 − 171. (in Chinese) [18] 于洪敏, 左景栾, 任韶然, 等. 注空气采油油井产出气体燃爆特性[J]. 中国石油大学学报(自然科学版), 2010, 34(6): 99 − 103.YU Hongmin, ZUO Jingluan, REN Shaoran, et al. Explosion characteristics of oil well produced gas by air injection for improved oil recovery[J]. Journal of China University of Petroleum, 2010, 34(6): 99 − 103. (in Chinese) [19] 赵利庆, 杨国骏, 李红波, 等. 注空气驱油工艺燃爆试验研究[J]. 油气田地面工程, 2019, 38(9): 12 − 17.doi:10.3969/j.issn.1006-6896.2019.09.003ZHAO Liqing, YANG Guojun, LI Hongbo, et al. Experimental study on explosion of air injection oil displacement process[J]. Oil-Gas Field Surface Engineering, 2019, 38(9): 12 − 17. (in Chinese)doi:10.3969/j.issn.1006-6896.2019.09.003 [20] LI P, LIU Z, LI M, et al. Investigation on the limiting oxygen concentration of combustible gas at high pressures and temperatures during oil recovery process[J]. Energy, 2021, 215: 119157.doi:10.1016/j.energy.2020.119157 -





 下载:
下载: